Your Location:Home > Products > 2-Aminobenzaldehyde
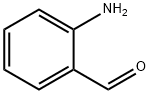

CAS No:529-23-7
MF:C7H7NO
Appearance:white or light-yellow crystalline powder
Specification:98.5%
UN number:-
Package:25kgs/drum
Supply Capacity:Tons
Product application:Pharmaceutical intermediates, Organic synthesis
Synonyms:2-Aminobenzaldehyde; Anthranilaldehyde; 2-amino-benzaldehyd;Benzaldehyde, 2-amino-;Benzaldehyde,2-amino-;O-AMINOBENZALDEHYDE;ANTHRANILALDEHYDE;2-Aminobenzaldeyhde;o-Amino Benzaldehyde 2-Amino Benzaldehyde;ORTHO-AMINOBENZALDEHYDE; 2-Aminobenzaldehyde,"2-Aminobenzaldehyde; 529-23-7; o-Aminobenzaldehyde; Anthranilaldehyde; EG769PG2AX; Ortho aminobenzaldehyde; DTXSID6060183; DTXCID4041317; 208-454-3; Benzaldehyde, 2-amino-; 2-amino-benzaldehyde; MFCD00007709; aminobenzaldehyde; 2-Formylaniline; O-amino benzaldehyde; anthranil aldehyde; EINECS 208-454-3; m-Amino benzaldehyde; aniline, 2-formyl-; Ambroxol Impurity 43; AI3-52264; UNII-EG769PG2AX; 2-ABA; SCHEMBL25504; SCHEMBL25505; 2-Aminobenzaldehyde, 98%; ghl.PD_Mitscher_leg0.1278; SCHEMBL10884849; SCHEMBL10897409; SCHEMBL14562109; SCHEMBL29776700; FXWFZIRWWNPPOV-UHFFFAOYSA-; BCP07330; BBL100092; SBB061591; STL280125; AKOS006221234; CS-W007486; FA71614; GS-3280; SY024955; DB-005184; NS00032615; ST51047883; EN300-118143; 10.14272/FXWFZIRWWNPPOV-UHFFFAOYSA-N.1; 2-Aminobenzaldehyde(Discontinued,See C4X-108543); doi:10.14272/FXWFZIRWWNPPOV-UHFFFAOYSA-N.1; Q21099244; 2-Formylaniline, Anthranilaldehyde, o-Aminobenzaldehyde; Z3034936011; InChI=1/C7H7NO/c8-7-4-2-1-3-6(7)5-9/h1-5H,8H2",121.140,C7H7NO,
Applications:
2-Aminobenzaldehyde, with CAS number 529-23-7, is a highly reactive aromatic intermediate characterized by the presence of both amino and formyl groups on adjacent positions of the benzene ring, typically appearing as a pale yellow crystalline solid or a low-melting mass. In terms of its detailed efficacy, this compound is renowned for its exceptional utility in organic synthesis, particularly as a fundamental building block in the Friedländer synthesis for the construction of functionalized quinoline derivatives, offering high chemical reactivity and precise regioselectivity in complex molecular assembly. Regarding specific applications, it is an indispensable raw material in the pharmaceutical industry for the development of diverse therapeutic agents, including antimalarial and anti-inflammatory drugs. Beyond medicine, it is extensively employed in the manufacturing of specialized dyes, fluorescent markers, and aromatic compounds, while also serving as a valuable reagent in advanced chemical analysis and laboratory research.